A STEP ABOVE
New treatment options for various diseases and rejuvenation of the human body using the body’s
fundamental building blocks, the “raw materials” from which cells are created. The Stem Cell.
This website is for educational purposes only. It offers health information and informs of some existing
alternative options. The site does not assume responsibility for the use of this material. We recommend
discussing the material with a medical professional well-versed in the subject.
What are Stem Cell?
They are undifferentiated cells without a specific function other than the ability to turn into cells with specialized functions based on the bodys’ requirements, but not all stem cells can change to just any cell and perform just any function.
Source of stem cell therapy?
They may come from various sources, such as adipose or fat tissue, placental tissue, umbilical cord tissue, or bone marrow.
How are Stem cells administered?
They can be administered intravenously (IV), directly into the spinal cord through intrathecal injection, directly injected into the affected areas, such as the face, hips, knees, or hands.
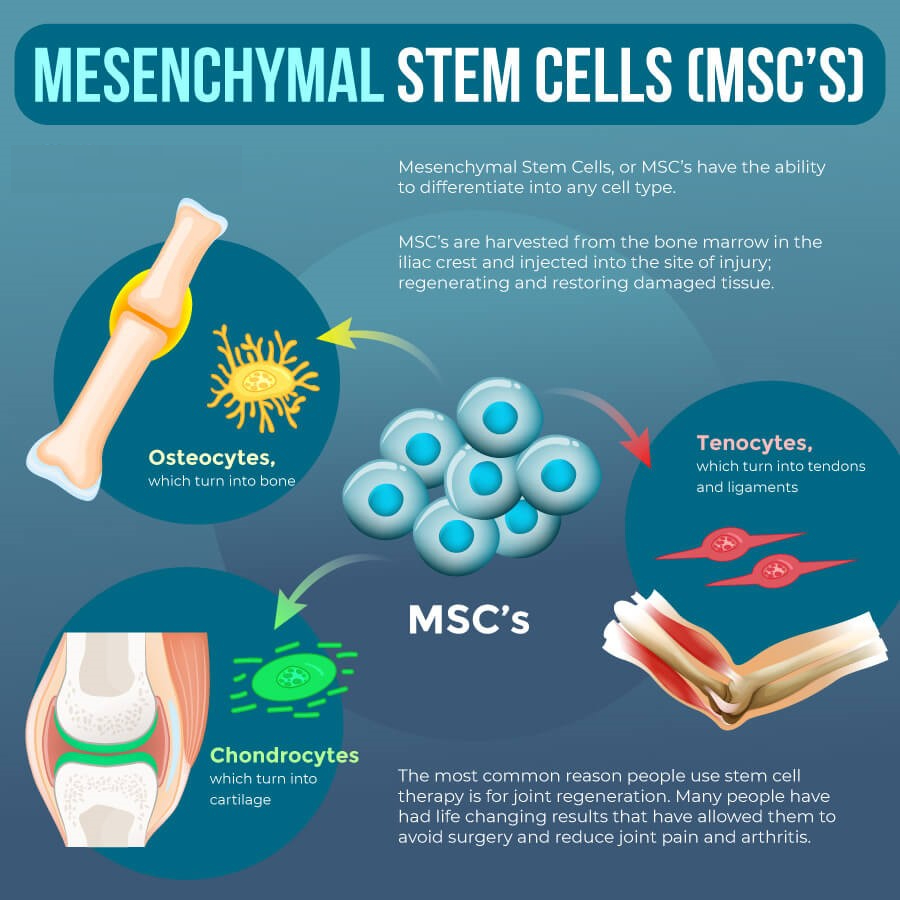
Mesenchymal Stem Cells (MSCs)
Mesenchymal stem cells (MSCs) originate from the connective tissue or stroma. around your body’s organs and tissues. Scientists have explored using them for new body tissues due to their ability to differentiate into any cell type.
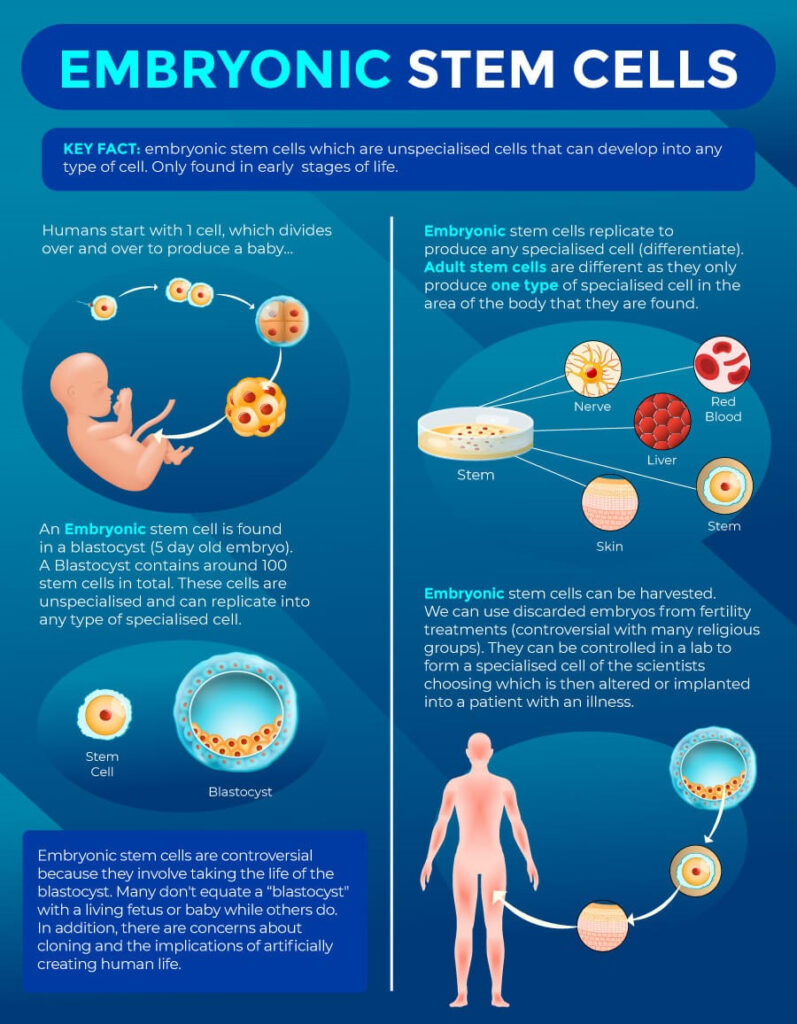
Embryonic Stem Cells
When a sperm fertilizes an egg, it forms an early-stage embryo called a blastocyst, which develops about 4 to 5 days after fertilization. At this stage, the blastocyst contains embryonic stem cells—unique cells capable of becoming many different types of cells, such as blood or skin cells. Because these cells are at an early stage of development, they have the potential to differentiate into a wider range of cell types compared to adult stem cells.
What is the controversy?
Human Embryonic Stem Cells may NOT be used and are NEVER used in any clinical setting, because it would destroy the blastocyst and thus a potential embryo. However, you may have heard of their use in some research settings from extra embryos from in vitro fertilization (IVF), which is highly controversial.
Source of cells for stem cell therapy?
Regenerative Medicine requires a reliable source of cell therapy in addition to biomaterial scaffolds and cytokine growth factors.
Umbilical Cord Cells – have been found to carry markers CD 34+ CD 45+ CD 73 and CD 90, which suggest the presence of stem cells, including HPC and MSCs, respectively in the product. These cells release Growth factors which play a critical role in repair. GF2 is involved in many aspects of growth, development and healing. It also plays a role in bone and cartilage formation, which may be beneficial for orthopedic injuries. The protein and IL-1, which is present in abundance in this product is a natural inhibitor of the pro-inflammatory state and plays a role in immuno-modulation inhibition of the pro inflammatory TNF– alpha. IL-1ra release from the cells may be critical in its ability to control the inflammatory response, which occurs, following disease and trauma states. The VEGF is a signaling protein produced by the stem cells that stimulate and vasogenesis and helps to restore oxygenation to tissues and cells when blood supplies in it is inadequate, vital in healing and repair. Other growth factors mediate cell, survival, migration, and proliferation.
ADIPOSE TISSUE – adipose tissue represents an abundant and accessible source of adult cells with the ability to differentiate along multiple lineage pathways. Stem cell therapy cells derived from adipose tissue are multipotent and promising for a range of therapeutic applications.
PERIPHERAL BLOOD – about 10,000 stem cell therapy cells can be obtained and of these 50% can become mesenchymal stem cell therapy cells.
BONE MARROW – approximately 50,000 NCP can be obtained from the sample. 50% become mesenchymal stem cell therapy cells.
LIPID TISSUES – about 5 to 10 million ASC can come from the sample. 90% become mesenchymal stem cell therapy cells.

Some of the Globally reported uses of Umbilical stem cell therapy

Stem Cell Based Therapy Information for Long Island, NY Residents
FAQs
What are stem cell–based therapies?
Stem cell–based therapies utilize biologic materials derived from stem cell sources to support regenerative signaling, inflammation modulation, and tissue support processes.
Are stem cell therapies FDA-approved?
Most stem cell–based therapies are not FDA-approved for clinical use outside of specific indications. Many applications are considered investigational and are offered in compliance with current regulations and informed consent.
Do stem cells turn into new tissue?
In most clinical applications, stem cell-related therapies work by supporting cellular communication and immune modulation, rather than directly becoming new tissue.
What conditions are stem cell-based therapies used for?
These therapies are commonly incorporated into regenerative care plans for joint support, recovery, and tissue health, depending on patient evaluation.
Who is a candidate for stem cell–based therapies?
Candidacy is determined through a comprehensive medical evaluation. Not all patients are appropriate candidates.
How Much Research Has Gone into Stem Cell Therapies?
Stem cell therapies have been the subject of extensive scientific research for several decades. Universities, research institutions, and medical centers across the United States — including those serving communities in Long Island, New York — continue to study how stem cells function and how they may be used in regenerative medicine.
The amount of research supporting a specific stem cell application depends on several factors, including the medical condition being studied and how long that particular approach has been under investigation. Some stem cell uses, such as bone marrow transplantation, have been researched for many years, while other applications are still considered experimental and are being evaluated through ongoing clinical trials.
Individuals in Long Island and surrounding areas who are exploring information about stem cell research are encouraged to review peer-reviewed studies and consult qualified healthcare professionals when considering medical decisions.
How Long Does It Take to See Results?
The timeline for potential results from stem cell–based therapies varies depending on the specific condition being studied, the type of stem cells involved, and the individual patient’s biological response. Research suggests that some regenerative processes may take weeks or months to develop, as tissue repair and cellular activity occur gradually over time.
For certain conditions under investigation — such as orthopedic, dermatologic, or hair-related applications — clinical studies often monitor outcomes over several months to evaluate effectiveness and safety.
Individuals in Long Island, New York, who are researching stem cell science should understand that many applications are still being evaluated in clinical trials. Anyone considering medical treatment should consult a licensed healthcare provider to discuss appropriate options, realistic timelines, and evidence-based expectations.
Is There Discomfort Associated with Stem Cell Therapies?
Reports of discomfort vary depending on the type of stem cell procedure being studied and the medical condition involved. In clinical research settings, some participants have reported temporary soreness, swelling, or sensitivity at the administration site. The intensity and duration of these symptoms differ from person to person.
Because stem cell applications are diverse and many are still under investigation, individuals in Long Island, New York, who are researching this field should consult licensed healthcare professionals for accurate information about potential side effects and recovery expectations related to any specific medical procedure
Why Do People Choose Stem Cells Over Other Treatments?
Stem cell science is an evolving area of regenerative medicine that researchers continue to study for its potential applications. Some individuals explore stem cell–based approaches because they are interested in emerging medical technologies currently being evaluated in clinical trials.
It is important to note that not all stem cell applications are FDA-approved, and many are still considered experimental. Residents of Long Island, NY, seeking information about stem cell research should review peer-reviewed studies and consult qualified medical professionals when comparing treatment options.
What are the Potential Risks of Stem Cell Therapies?
As with any medical intervention under study, stem cell–based treatments may carry potential risks. Reported concerns in medical literature can include infection, immune reactions, unintended tissue growth, or lack of effectiveness. The level of risk depends on the type of stem cell used, how it is processed, and how it is administered.
Because research is ongoing, individuals in Long Island and surrounding communities are encouraged to rely on credible scientific sources and speak directly with licensed healthcare providers to fully understand the potential benefits and risks associated with any medical procedure.
Universal Patient Disclosure
All regenerative therapies described are not guaranteed and are not FDA-approved to diagnose, treat, cure, or prevent disease. Individual results may vary. All treatments are provided under physician supervision following a comprehensive medical evaluation and informed consent.